BREAKING: Johnson & Johnson Stock Faces 10% Correction Warning as FDA Decisions Loom
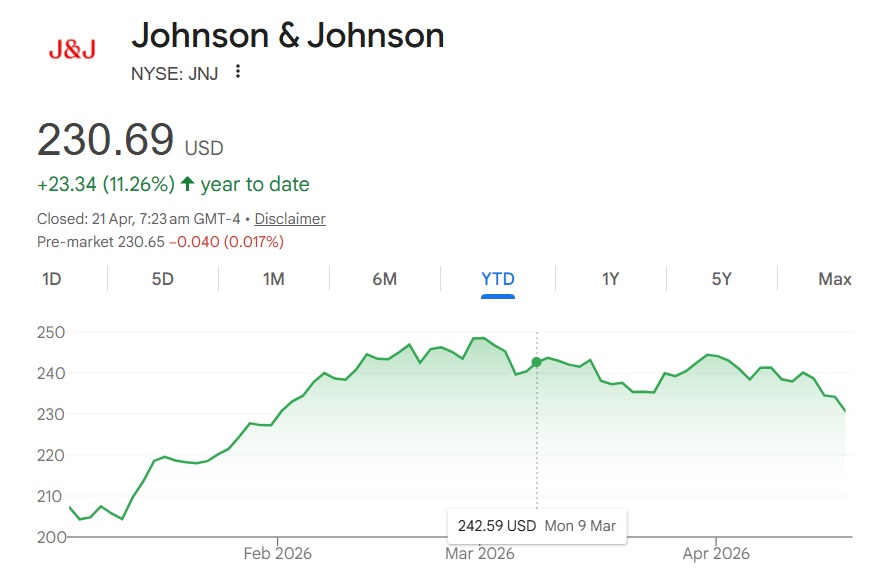
Johnson & Johnson (NYSE: JNJ) is bracing for potential volatility as analysts warn of a 10% stock price correction ahead of two critical upcoming FDA decisions. The pharmaceutical giant's shares, which surged nearly 11.5% year-to-date on the back of recent approvals for Icotyde and Rybrevant, now face a pivotal test with new regulatory verdicts on the horizon that could reshape its market valuation.
 Source: Google
Source: Google
However, 2 other FDA approvals are in line that can make or break Johnson & Johnson’s stock price. The pharmaceutical company has submitted one for review with the FDA, and the other still remains in the testing phase. In this article, we will explain the upcoming approvals and how they will help JNJ stock surge in value.
Johnson & Johnson Stock: 2 FDA Decisions Hold the Key For JNJ To Rally

The 2 drugs in process for an FDA nod from Johnson & Johnson are:
is a robotic surgery system and is currently under FDA review, needing regulatory clearance. This is the major catalyst, as it involves the MedTech system. The pharma giant submitted the trials and documents in January 2026 to the department. Investors are waiting for the FDA’s decision, and a ‘yes’ would give Johnson & Johnson stock the much-needed boost in the charts.
is a combination therapy for gut health and is currently in clinical trials. The testing and research phase is ongoing, and it is reported that the company might submit it to the FDA in 2027. If this receives the nod, Johnson & Johnson stock could be set for the long term in 2027 and beyond. While Ottava’s approval can set JNJ in motion in 2026, JNJ-78934804’s approval can continue the momentum in 2027. This fills the gap on both short-term and long-term profits from the pharma giant.


Log in to Reply
Log in to comment your thoughtsComments
Related Articles
|Square
Get the BTCC app to start your crypto journey
Get started today Scan to join our 100M+ users